#16. Go With Your Gut (Biome)
In which the E@L experiences the symptoms of antibiotic-induced dysbiosis
First, I want to apologize to my regular readers. I have been offline for two months because I was sick. I don’t know what with, or why. At risk of boring you with my ailments, a quick review is necessary to introduce today's topic. What happened to me is probably not as important as how the medical establishment responded, how I responded, and what it can teach us about the intersection between environment, our bodies, and medicine.
In late July I found a tick embedded in my leg that had to be removed by outpatient surgery, followed by a two-week course of doxycycline. I’ve had Lyme disease before; it hit me like a truck, with chills, fever, body pains and a bullseye on my abdomen. This time I had no symptoms. But three weeks later, I started experiencing chills and fever, so my GP put me on a second course of doxycycline. Within a week, I was violently nauseous, which garnered a prescription for methyl-prednisolone, a strong steroid. That induced double vision, vertigo, and severe abdominal cramps, leading to a trip to the ER and a prescription for anti-cramping drugs (docylamine). This caused extreme constipation, necessitating the use of laxatives.
Meanwhile I had lost my taste buds and appetite, as well as fifteen lbs. Add brain fog, dizziness, hand tremors, and general weakness to the list. As if that wasn’t enough, I had a relapse of sciatica, which was supposedly cured by back surgery three years ago. Now I can’t walk, can’t eat, am extremely weak, and can’t, well, you know. At this point I decided that enough was enough. I stopped taking drugs and went au natural. Despite my lack of appetite, I loaded up on high fiber foods, smoothies, and prune juice. It took about two weeks to get the drugs out of my system, and another month to re-establish normal digestive function, with the assistance of OTC pro-biotics.
Antibiotic means what it sounds like
Lyme disease is caused by bacteria, specifically the spirochete Borreliella burgdorferi (and some other closely related species including Ehrlichia, Babesia, and Rickettsia, for rocky mountain spotted fever). Because of the difficulty in identifying the specific culprit, these diseases are typically treated with wide-spectrum Tetracycline-class antibiotics such as doxycycline and amoxicillin.
These drugs can cause significant changes in the microflora of the human gut (the gut biome). The goal of using such antibiotics is typically to stop abnormal growth of bacteria, such as the type that cause Lyme disease. However, doxycycline and other drugs not only prevent growth of invasive pathogens, but actually kill resident bacteria.
The gut surface is the largest interface (200-400 m2) between our bodies and the environment. The bacteria in our gut outnumber the cells in our bodies by a factor of 10, and their collective DNA exceeds our own by a factor of 100. Intestinal bacteria play a crucial role in maintaining our immune systems and metabolic homeostasis and protecting against pathogens. Gut bacteria are essential for digestion of many foods, absorption of nutrients, synthesize necessary vitamins, and cause fermentation of some food types. They also provide necessary protection from the growth of foreign microbes.
Dysbiosis – Not your average biosis
Dysbiosis is the disturbance of the quantity, variety and/or location of microorganisms on or within the human meta-organism. In the digestive system, dysbiosis causes loss of beneficial bacteria, overgrowth of potentially pathogenic bacteria, and loss of overall bacterial diversity. Antibiotics significantly reduce gut bacteria, and in certain patients, these medicines completely eliminate specific bacterial communities.
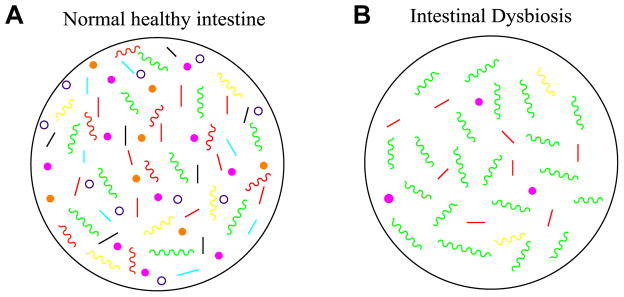
The widespread use of tetracycline antibiotics, especially doxycycline, is a major cause of dysbiosis. At low dosage, doxycycline has little effect on the gut microbiota with the exception of Enterococci and E. coli, but at higher doses it reduces the abundance of most normal gut flora. The abundance of Bacteroides sp. and Firmicutes sp were significantly reduced in endocarditis patients treated with doxycycline for <3 months and >3 months, respectively, compared to control (non-sick) patients. At normal dosage (100-200 mg/day), doxycycline markedly reduced the diversity of Bifidobacteria populations in one study and eliminated Fusobacterium sp in another. Recovery of normal gut populations required anywhere from 9 to 28 days.
Addition of glucose to the diet of mice (which normally consume high fiber diets), increased the susceptibility of Bacteroides to amoxicillin. If we were to extend this to humans, we might conclude that a diet that is high in carbohydrates and low in fiber (typical of our society) would make us more susceptible to antibiotic-induced disturbance of the gut biome. But that may be a stretch.
Let me be your Sledgehammer
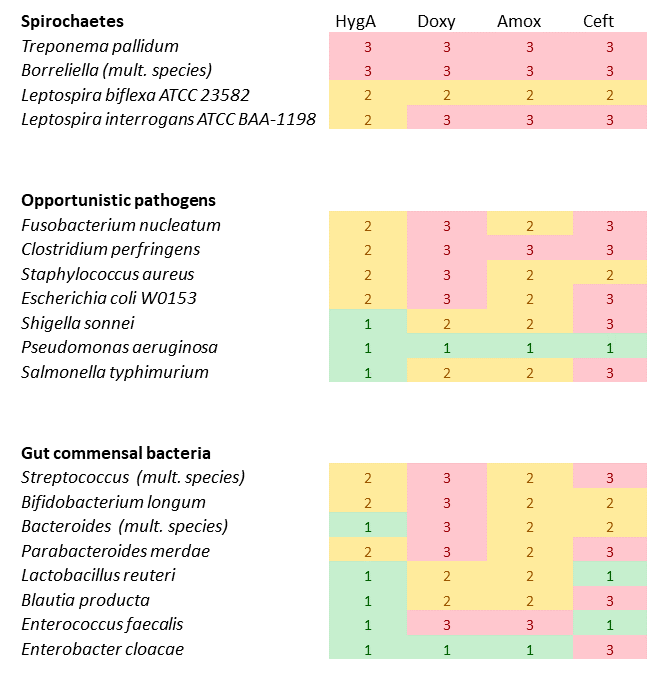
Doxycycline has been described as "a pretty general sledgehammer". In contrast, a rarely used treatment for Lyme disease is hygromycin A, which is a much more specific antagonist of Boreliella and other spirochaetes, and much less active against common gut biota (Table 1). Looking at minimum inhibitory concentrations (MIC, the lowest amount needed to inhibit bacterial growth, in µg/mL), hygromycin was as effective as doxycycline (MIC < 0.25) against most Lyme-disease associated bacteria, but much less active (MIC 1-32) against opportunistic pathogens such as Fusobacterium, Clostridium, Staphylococcus, or E. coli, and virtually useless (MIC>64) against Shigella and Pseudomonas. But whereas doxycycline and amoxicillin were highly toxic to normal gut bacteria, hygromycin had low impacts on Streptococcus salivarius, Bifidobacterium longum, Bacteroides sp., or Lactobacillus reuteri (MIC 2-32), and virtually no impact (MIC>64) on Enterococcus faecalis, or Enterobacter cloacae.
Table 1. Effectiveness of four antibiotics against digestive tract bacteria. Red boxes (value 3) indicate strong antibiotic activity, whereas yellow (2) or green (1) indicate lower antibiotic activity. HygA=Hygromycin A; Doxy=Doxycycline; Amox=Amoxicillin; Ceft=Ceftriaxone (Data adapted from Table 1 of Liemer et al., 2021)
Dysbiosis can lead to many physiological problems. These include inflammatory bowel diseases (IBD), irritable bowel syndrome (IBS), diabetes, obesity, cancer, cardiovascular and central nervous system disorders. In studies on both humans and animals, obesity seems to be associated with a decrease in Bacteroides and an increase in Firmicutes sp. Insulin-dependent diabetes melitus (IDDM) is associated with a decrease in Bifidobacteria, Lactobacillus, and Prevotella, and an increase in Bacteroides and Clostridium, although it is unclear if dysbiosis is a causative factor or an effect of diabetes. Various human and animal studies, however, suggest that changes in the microbiota may precede the onset of IDDM. There are also strong implications that intestinal dysbiosis may play a role in the pathogenesis of Autism Spectrum Disorders. Intestinal dysbiosis has also been linked to colorectal cancer, the third most common cancer and second leading cause of cancer death.
Dysbiosis may also lead to immune system activation and subsequent systemic inflammation, neuroinflammation and changes in central nervous system functioning and behavior. In fact, there is a direct relationship between the gut microbiome and brain function, mediated by molecules that travel via the peripheral nervous system. A review of the impacts of dysbiosis due to antibiotic administration demonstrated that it slows the recovery of nerve damage, such as sciatic compression, whereas administration of probiotics appeared to promote regeneration in sciatic nerves. A high fat diet in mice (again) caused dysbiosis that was associated with increased peripheral neuropathy (don’t ask me how they know this), and that reversal of the diet to a normal (for mice) diet reduced both dysbiosis and peripheral nerve pain (I don’t even want to know). Could this explain the onset of my sciatica-related neuropathy? It’s an enticing idea, but maybe a stretch.
Another side effect of dysbiosis is mechanical allodynia, i.e a painful sensation caused by innocuous stimuli like light touch. Strangely, one of my lingering symptoms has been sensitivity of my ribcage to touch or even clothing.
Lessons learned?
I still don't know what disease I had; I was tested for five tick-borne diseases and multiple viruses at three-week intervals, all with negative results. I went down myriad internet rabbit holes trying to sleuth out the basis of my misery and never found it. Nor am I likely to, despite the fact that I still have lingering, possibly permanent bodily changes.
But I think I finally understand what happened to me. In response to the perceived onset of Lyme disease, I was prescribed a wide-spectrum antibiotic twice within three weeks (which seemed like the responsible choice at the time). But without any actual pathogenic bacteria for doxycycline to fight, its considerable antibiotic power was unleashed against my normal, beneficial, gut microbiota. This had immediate detrimental impacts on my gut biome, which probably precipitated my nausea, cramps, constipation, and other symptoms. It’s plausible that this also led to a flare-up of neuropathic pain associated with my previous sciatica. I’m now taking probiotics (which include several strains of Bifidobacteria and Lactobacillus) in order to counteract the devastation this caused to my digestive system.
The widespread administration of wide-spectrum antibiotics such as doxycyline can cause many other side effects and problems. As a society, we should want to discourage the use of such “sledgehammer” type drugs. But that might be a difficult fight because they are so effective. From a personal perspective, I think the best course of action is just to be forewarned. Do not take these antibiotics unless you are prepared for the consequences, and make sure you balance them with a counteractive intake of probiotics. When it comes to drug prescriptions, go with your gut.